What is an Electronic Nose?
An electronic nose is a device that has the ability to detect a multitude of different gases and determine their concentrations. It is often referred to as a bionic or biomimetic device, as it is a design inspired by the human nose. It achieves this function by assigning an electronic "fingerprint" to each gas. When the device is put in the presence of a mixture of gases, it uses a series of different gas sensors to detect the fingerprint of all the gases, and then uses these fingerprints to determine how much of each gas is present.
Our device works specifically on the principle of amperometric sensing. That is, it utilizes sensors composed of semiconductors whose electrical conductivity (G) changes with the concentration of gas present. Each sensor is made from a different semiconductor material and each responds differently to any given gas. For example, four different sensors will have a different change in conductivity with a changing concentration of some gas (ie. Gas A).
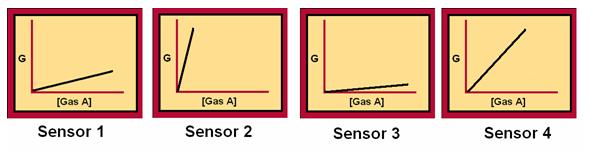
This is where the concept of a fingerprint comes from. No other gas other than Gas A will cause these specific slopes in all four of the above graphs. As a result, the slope of these curves represents the unique fingerprint of Gas A.
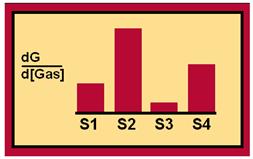 Gas A Fingerprint
Gas A Fingerprint
These sensors are cross-reactive, and so any given sensor will experience a change in conductivity from every gas present. Hence, in a mixture of gases, each gas present will affect each sensor, leading to a measured fingerprint that is the convolution of all of the fingerprints of the individual gases. With a prior knowledge of each gas's fingerprint (gained from calibration), and using linear algebra based algorithms, the fingerprint can be broken down into a linear superposition of the "basis" fingerprints, hence allowing the electronic nose to determine the identity and concentration of each gas in the mixture.
How do the Sensors Work?
The sensors utilize a sensing mechanism that works on a nanoscale. They are composed of metal-oxide nanoparticles that have semiconducting properties. When exposed to a gas, the gas molecules will chemisorb to the surface of the nanoparticles, and through various reduction reactions, donate an electron into the conduction band of the nanopartcile material. The electron usually penetrates on the order of about 3 nm into the material. This creates a "space charge" layer that is 3 nm deep, in which the semiconductor has a higher electrical conductivity. Hence, over the entire material, the conductivity rises as the concentration of the gas increases. Because the space-charge layer is only about 3 nm, the particles have to be very small (less than ~ 100 nm), before the 3 nm space-charge layer becomes significant, and so to exploit this transduction phenomenon, nanoparticles must be used.
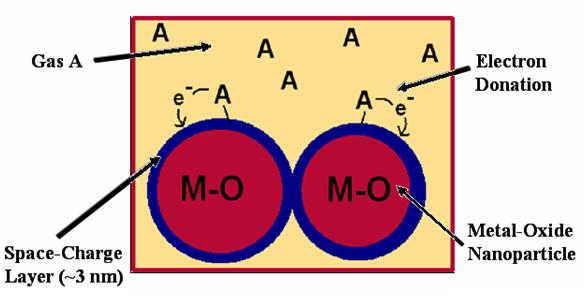 Method of Transduction
Method of Transduction
The reaction between the gas and the nanoparticle depends on the identity of the gas and the identity of the metal oxide that composes the nanoparticles. Hence, the chemistry changes for each gas and each sensor, and as such, so does the conductivity curve.
An Assembled Device
The final device consists of two main components. The first is a microheater, which is included due to the fact that temperatures in the range of 100-500°C are required for these chemical reactions to occur. The microheater is able to heat the local area of the chip using very little power, and without perturbing other sensors in the vicinity. The second major component is the microfabricated electrode, on top of which the nanoparticle material is deposited by spin coating. Below is an example of an electrode/sensing layer and a microheater. The two devices are separated by a thin SiO2 layer, allowing thermal conductivity and electrical insulation between the heater and the sensing layer above it. The entire device is fabricated on a silicon substrate with a deposited SiO2 layer.
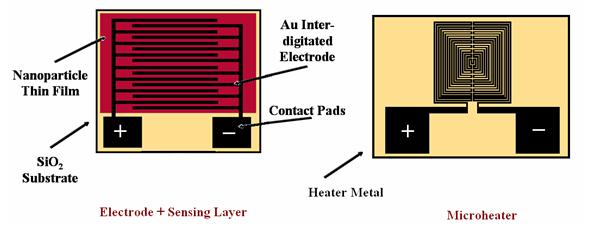
Each device has 4 connections, two terminals for the electrode and two for the heater. The electrodes of each sensor are assembled into a current divider circuit, where the current in each branch gives the conductivity of the sensor and hence, the current ratio of all of the branches gives the electronic fingerprint. The heater is controlled using a feedback loop, based on the heater-metal's own resistivity being a function of temperature.
Noise Control
Our design includes a special feature to quantify the noise in the measurement. By using more sensors than gases present, the deconvolution of the electronic fingerprint represents an "overdetermined system". As such, the measured resistance of each sensor plotted in an N-dimensional "sensor space" will give a point that lies out of the (N-1)-dimensional space defined by the N basis vectors of each gas's fingerprint. The magnitude of the vector connecting the point to this space represents the error in the measurement, and is easily measurable. Additionally, our sensing algorithm can choose which subset of sensors create the smallest error vector and use these in the deconvolution algorithm, so as to maximize the signal-to-noise ratio.

