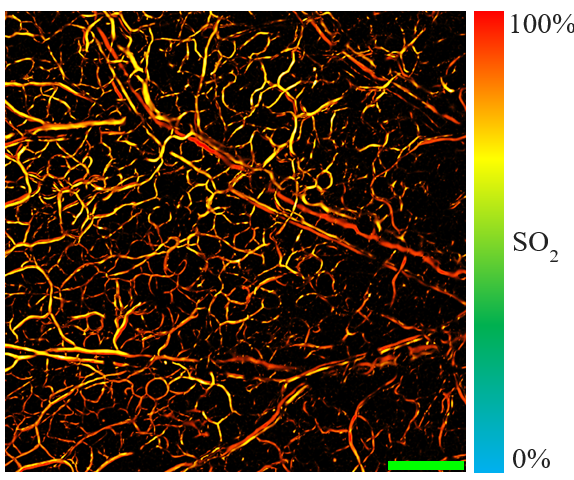
Our lab is interested in designing and developing novel optical systems for clinical and pre-clinical biomedical applications. These new technologies aim to provide clinicians and researchers with novel capabilities and information that is presently difficult to obtain with existing techniques. Some of our research interests are including
- Photoacoustic remote sensing (PARS) microscopy
- Multi-scale functional and molecular photoacoustic imaging
- Non-contact detection of photoacoustic signals
- All optical nano-structured detectors for photoacoustic tomography
- Photoacoustic wavefront shaping
- Optical coherence tomography (OCT)
- Multi-photon microscopy
- Novel multi-modality optical imaging techniques
- Design and fabrication of micro-endoscopy systems
- Super-resolution imaging techniques
- Real-time and 3D biomedical imaging
- Dual-Modality photoacoustic and ultrasound imaging systems
- Novel optical and fiber-based components
- Novel laser systems for biomedical applications
- Optical sensors
Professor Parsin Haji Reza has pioneered photoacoustic remote sensing (PARS) microscopy. Currently, our primary research focus is on the development of deep cellular resolution PARS microscopy techniques. We are working on hardware and software optimization as well as phantom and in vivo studies to enable the first in human test in near future. Our secondary research vision involves the development of multi-modality optical imaging techniques as well as novel all-optical detection methods for photoacoustic tomography. Some of the main applications that we are currently pursuing are including:
Eliminating Positive Surgical Margins
Achieving negative surgical margins is one of the fundamental principles of surgical oncology. It can prove challenging in many oncologic surgeries, including breast conserving partial mastectomies, resections for head and neck cancer, rectal cancer, genitourinary cancer, and malignant intracranial tumors. It is critically important to achieve a final pathology specimen with a rim of normal tissue around the tumor. This assures the oncologic care team that all resectable disease has been taken out, and no malignant tissue is left behind. In multiple clinical trials, surgical margins have proven to be strongly associated with a patient’s disease-free survival and overall survival. The consequences of not achieving adequate surgical margins include: poorer patient outcomes, follow up operations and re-excisions to achieve negative margins, and the need for potentially unnecessary additional treatments such as adjuvant chemotherapy and/or radiotherapy.
To address this, contemporary surgical oncologists have relied heavily on clinical judgment and histopathologic identification of margins to guide them. Increasing use of intraoperative frozen pathology also aids in decreasing the rate of positive margins. Although intraoperative frozen pathology has proven to be a powerful tool that decreases margin positive rates and reoperation rates, the accuracy of the frozen tissue section analysis compared to final pathologic analysis can be variable. Furthermore, it takes about one hour to perform one frozen pathologic assessment of margins, which significantly prolongs the operating time and consequently time and cost of the operation, as well as increased anesthesia risks for the patients. In certain oncologic surgeries where multiple frozen pathology analysis is required, this adds a significant burden to the healthcare system and increases the complication rate for patients.
Attempts to improve this intraoperative margins analysis have mainly focused on better surgical techniques, reliance on neoadjuvant chemotherapy or radiotherapy, or emerging fluorescence-based tumor margin analysis technology. These approaches do not come yet close to the gold standard of microscopy-based histopathologic analysis. Thus the best proactive step to mitigate margin positive rates is still intraoperative frozen pathology, despite its drawbacks.
There is a need for a new technique to provide rapid, repeatable, real-time, non-contact microscopy level imaging to be attained for pathologic analysis intra-operatively. Because of the non-contact approach, and high level of resolution achievable, our currently discovered technology can be a great solution for imaging the margins of a surgical cavity to check for any malignant cells reaching the margins. Unlike any other histopathologic tool, this analysis can be done with the tissue in question still in situ, and pathologic analysis can be done in a matter of seconds in real-time rather than taking up to an hour with the current standard of frozen pathology. The result would be: dramatically shortened operation times, increased throughput of surgeries per day per operating room, superior oncologic outcomes similar to frozen pathology, and decreased anesthetic complications for the patient. These significant advantages make sense from a patient care and cancer care point of view as well as from a health economics of point of view, which will encourage greater use of intraoperative pathologic analysis for everyone’s benefit.
Angiogenesis imaging:
A tumor, in order to grow and reach the metastatic spread phase, needs to develop an independent blood network to supply nutrients and oxygen and to remove waste products. The growth of these abnormal blood vessels is called angiogenesis. Angiogenesis plays a significant role in tumor growth and metastasis. Tumor growth and metastasis are responsible for more than 90% of cancer death. As a result, angiogenesis has become one of the heavily funded areas of medical research in history. Because haemoglobin provides ~ 2 orders of magnitude higher optical absorption compared to surrounding tissues, photoacoustic imaging is one of the best solutions for imaging blood vessels and angiogenesis. In our lab we are working on longitudinal structural and functional angiogenesis studies. One of primary research direction is to test and analyze the angiogenesis inhibitor effectiveness by novel imaging techniques.
Ophthalmology:
Nearly 1 in 3 individuals will experience some degree of vision loss by the age of 65. Our vision is a central component of perception and interaction in our world, which can make vision loss an extremely debilitating process for many. This has serious implications for a patient’s quality of life as well as the cost to society for health care needs. The majority of these age-related vision loss is due to Age-related macular degeneration (AMD), diabetic retinopathy, glaucoma, as well as other disease processes. This has brought about the adoption of advanced modern imaging techniques such as Optical coherence tomography (OCT), fluorescence-based intraocular imaging, and ultrasound-based imaging, with the aim being a better characterization of the vasculature in the hopes of detecting and treating these disease processes early. We recently pioneered a non-contact non-invasive optical absorption based imaging technique (PARS Microscopy). This technique enables ultra sensitive and accurate structural and functional imaging of intraocular vasculatures. PARS technology provides a direct accurate measurement of oxygen saturation (sO2) which is essential information for early detection and understanding the age-related vision loss problems. Our group is working to customize and utilize the PARS microscopy to be used as an add-on technology to the currently available imaging systems in clinical ophthalmology such as OCT.
Non-contact Biopsies for Endoscopy
Endoscopy remains the gold standard technique for finding and assessing multiple types of cancers residing in internal organs and structures. This includes all gastrointestinal cancers, all head and neck cancers, most lung cancers, bladder cancers, cervix and vaginal cancers, as well as laproscopic and thorascopic evaluations for malignant disease. The real benefit of endoscopy is that it allows a minimally invasive approach to see inside cavities and obtain tissue specimens (biopsies) that allow histopathologic confirmation of cancers. The sensitivity, specificity, and ability to obtain tissue specimen makes endoscopy irreplaceable in its role in cancer care. Although advances in imaging have been suggested as an alternative to endoscopy, there are still significant short-comings that relegate non-invasive imaging to being used as a second-line approach or as an adjunct to endoscopy. In our lab, we are developing optical endoscopy techniques to provide functional and molecular marker imaging, which allows in situ diagnosis and characterization of cancer’s microenvironment (ex: oxygenation status), aggressiveness, and sensitivity to targeted therapies.
- Photoacoustic remote sensing (PARS) microscopy
- Multi-scale functional and molecular photoacoustic imaging
- Non-contact detection of photoacoustic signals
- All optical nano-structured detectors for photoacoustic tomography
- Photoacoustic wavefront shaping
- Optical coherence tomography (OCT)
- Multi-photon microscopy
- Novel multi-modality optical imaging techniques
- Design and fabrication of micro-endoscopy systems
- Super-resolution imaging techniques
- Real-time and 3D biomedical imaging
- Dual-Modality photoacoustic and ultrasound imaging systems
- Novel optical and fiber-based components
- Novel laser systems for biomedical applications
- Optical sensors
Professor Parsin Haji Reza has pioneered photoacoustic remote sensing (PARS) microscopy. Currently, our primary research focus is on the development of deep cellular resolution PARS microscopy techniques. We are working on hardware and software optimization as well as phantom and in vivo studies to enable the first in human test in near future. Our secondary research vision involves the development of multi-modality optical imaging techniques as well as novel all-optical detection methods for photoacoustic tomography. Some of the main applications that we are currently pursuing are including:
Eliminating Positive Surgical Margins
Achieving negative surgical margins is one of the fundamental principles of surgical oncology. It can prove challenging in many oncologic surgeries, including breast conserving partial mastectomies, resections for head and neck cancer, rectal cancer, genitourinary cancer, and malignant intracranial tumors. It is critically important to achieve a final pathology specimen with a rim of normal tissue around the tumor. This assures the oncologic care team that all resectable disease has been taken out, and no malignant tissue is left behind. In multiple clinical trials, surgical margins have proven to be strongly associated with a patient’s disease-free survival and overall survival. The consequences of not achieving adequate surgical margins include: poorer patient outcomes, follow up operations and re-excisions to achieve negative margins, and the need for potentially unnecessary additional treatments such as adjuvant chemotherapy and/or radiotherapy.
To address this, contemporary surgical oncologists have relied heavily on clinical judgment and histopathologic identification of margins to guide them. Increasing use of intraoperative frozen pathology also aids in decreasing the rate of positive margins. Although intraoperative frozen pathology has proven to be a powerful tool that decreases margin positive rates and reoperation rates, the accuracy of the frozen tissue section analysis compared to final pathologic analysis can be variable. Furthermore, it takes about one hour to perform one frozen pathologic assessment of margins, which significantly prolongs the operating time and consequently time and cost of the operation, as well as increased anesthesia risks for the patients. In certain oncologic surgeries where multiple frozen pathology analysis is required, this adds a significant burden to the healthcare system and increases the complication rate for patients.
Attempts to improve this intraoperative margins analysis have mainly focused on better surgical techniques, reliance on neoadjuvant chemotherapy or radiotherapy, or emerging fluorescence-based tumor margin analysis technology. These approaches do not come yet close to the gold standard of microscopy-based histopathologic analysis. Thus the best proactive step to mitigate margin positive rates is still intraoperative frozen pathology, despite its drawbacks.
There is a need for a new technique to provide rapid, repeatable, real-time, non-contact microscopy level imaging to be attained for pathologic analysis intra-operatively. Because of the non-contact approach, and high level of resolution achievable, our currently discovered technology can be a great solution for imaging the margins of a surgical cavity to check for any malignant cells reaching the margins. Unlike any other histopathologic tool, this analysis can be done with the tissue in question still in situ, and pathologic analysis can be done in a matter of seconds in real-time rather than taking up to an hour with the current standard of frozen pathology. The result would be: dramatically shortened operation times, increased throughput of surgeries per day per operating room, superior oncologic outcomes similar to frozen pathology, and decreased anesthetic complications for the patient. These significant advantages make sense from a patient care and cancer care point of view as well as from a health economics of point of view, which will encourage greater use of intraoperative pathologic analysis for everyone’s benefit.
Angiogenesis imaging:
A tumor, in order to grow and reach the metastatic spread phase, needs to develop an independent blood network to supply nutrients and oxygen and to remove waste products. The growth of these abnormal blood vessels is called angiogenesis. Angiogenesis plays a significant role in tumor growth and metastasis. Tumor growth and metastasis are responsible for more than 90% of cancer death. As a result, angiogenesis has become one of the heavily funded areas of medical research in history. Because haemoglobin provides ~ 2 orders of magnitude higher optical absorption compared to surrounding tissues, photoacoustic imaging is one of the best solutions for imaging blood vessels and angiogenesis. In our lab we are working on longitudinal structural and functional angiogenesis studies. One of primary research direction is to test and analyze the angiogenesis inhibitor effectiveness by novel imaging techniques.
Ophthalmology:
Nearly 1 in 3 individuals will experience some degree of vision loss by the age of 65. Our vision is a central component of perception and interaction in our world, which can make vision loss an extremely debilitating process for many. This has serious implications for a patient’s quality of life as well as the cost to society for health care needs. The majority of these age-related vision loss is due to Age-related macular degeneration (AMD), diabetic retinopathy, glaucoma, as well as other disease processes. This has brought about the adoption of advanced modern imaging techniques such as Optical coherence tomography (OCT), fluorescence-based intraocular imaging, and ultrasound-based imaging, with the aim being a better characterization of the vasculature in the hopes of detecting and treating these disease processes early. We recently pioneered a non-contact non-invasive optical absorption based imaging technique (PARS Microscopy). This technique enables ultra sensitive and accurate structural and functional imaging of intraocular vasculatures. PARS technology provides a direct accurate measurement of oxygen saturation (sO2) which is essential information for early detection and understanding the age-related vision loss problems. Our group is working to customize and utilize the PARS microscopy to be used as an add-on technology to the currently available imaging systems in clinical ophthalmology such as OCT.
Non-contact Biopsies for Endoscopy
Endoscopy remains the gold standard technique for finding and assessing multiple types of cancers residing in internal organs and structures. This includes all gastrointestinal cancers, all head and neck cancers, most lung cancers, bladder cancers, cervix and vaginal cancers, as well as laproscopic and thorascopic evaluations for malignant disease. The real benefit of endoscopy is that it allows a minimally invasive approach to see inside cavities and obtain tissue specimens (biopsies) that allow histopathologic confirmation of cancers. The sensitivity, specificity, and ability to obtain tissue specimen makes endoscopy irreplaceable in its role in cancer care. Although advances in imaging have been suggested as an alternative to endoscopy, there are still significant short-comings that relegate non-invasive imaging to being used as a second-line approach or as an adjunct to endoscopy. In our lab, we are developing optical endoscopy techniques to provide functional and molecular marker imaging, which allows in situ diagnosis and characterization of cancer’s microenvironment (ex: oxygenation status), aggressiveness, and sensitivity to targeted therapies.
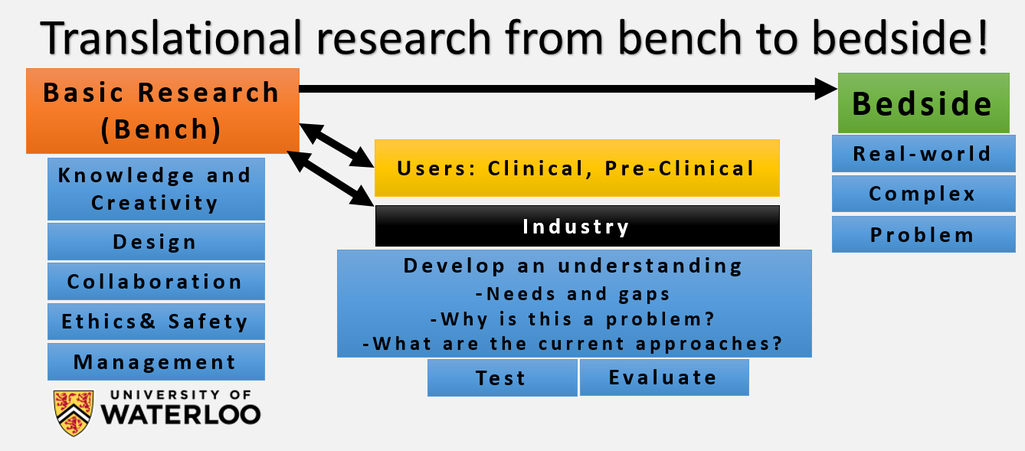
Our Research philosophy:
Listening to the Sound of Light: Biomedical Photoacoustic Imaging:
Photoacoustic can be defined as listening to the sound of light. Discovered by Alexander Graham Bell in 1880, the photoacoustic effect involves the absorption of laser light and subsequent conversion to sound waves. However, recently photoacoustic has became a popular field and it is utilized for biomedical imaging. By taking advantage of the photoacoustic effect, anything that absorbs light including RNA, DNA, lipid, blood, and melanin (the pigment in the skin), will generate an ultrasound wave. Photoacoustic imaging is the only imaging modality that provides high-resolution optical absorption imaging contrast, allowing it to be used for novel pre-clinical and clinical applications in oncology, dermatology, neurology, as well as many other disciplines. One significant application for photoacoustic imaging is imaging of angiogenesis (the growth of new blood vessels). A tumor, in order to grow and reach the metastatic spread phase, needs to develop an independent blood network to supply nutrients and oxygen and to remove waste products. Angiogenesis plays a significant role in tumor growth and metastasis. Tumor growth and metastasis are responsible for more than 90% of cancer death. As a result, angiogenesis has become one of the most heavily funded areas of medical research in history. Since blood absorbs light very strongly compared to surround tissues, photoacoustic imaging provides one of the best solution for imaging blood vessels and angiogenesis.
Photoacoustic can be defined as listening to the sound of light. Discovered by Alexander Graham Bell in 1880, the photoacoustic effect involves the absorption of laser light and subsequent conversion to sound waves. However, recently photoacoustic has became a popular field and it is utilized for biomedical imaging. By taking advantage of the photoacoustic effect, anything that absorbs light including RNA, DNA, lipid, blood, and melanin (the pigment in the skin), will generate an ultrasound wave. Photoacoustic imaging is the only imaging modality that provides high-resolution optical absorption imaging contrast, allowing it to be used for novel pre-clinical and clinical applications in oncology, dermatology, neurology, as well as many other disciplines. One significant application for photoacoustic imaging is imaging of angiogenesis (the growth of new blood vessels). A tumor, in order to grow and reach the metastatic spread phase, needs to develop an independent blood network to supply nutrients and oxygen and to remove waste products. Angiogenesis plays a significant role in tumor growth and metastasis. Tumor growth and metastasis are responsible for more than 90% of cancer death. As a result, angiogenesis has become one of the most heavily funded areas of medical research in history. Since blood absorbs light very strongly compared to surround tissues, photoacoustic imaging provides one of the best solution for imaging blood vessels and angiogenesis.
Where to start?
1- Lihong V Wang et at. "A practical guide to photoacoustic tomography in the life sciences" Nature Methods, Vol.13 No.8, 2016, doi:10.1038/nmeth.3925
2- Yong Zhou et. al, “Tutorial on photoacoustic tomography,” J. Biomed. Opt. 21(6), 061007 (2016), doi: 10.1117/1.JBO.21.6.061007.
3- Junjie Yao et. al, "Sensitivity of photoacoustic microscopy", Photoacoustics 2 (2014) 87–101.
4- Paul Beard, "Biomedical photoacoustic imaging" Interface Focus. 2011 Aug 6; 1(4): 602–631.
5- Vasilis Ntziachristos "Going deeper than microscopy: the optical imaging frontier in biology", Nature Methods 7, 603–614 (2010)
1- Lihong V Wang et at. "A practical guide to photoacoustic tomography in the life sciences" Nature Methods, Vol.13 No.8, 2016, doi:10.1038/nmeth.3925
2- Yong Zhou et. al, “Tutorial on photoacoustic tomography,” J. Biomed. Opt. 21(6), 061007 (2016), doi: 10.1117/1.JBO.21.6.061007.
3- Junjie Yao et. al, "Sensitivity of photoacoustic microscopy", Photoacoustics 2 (2014) 87–101.
4- Paul Beard, "Biomedical photoacoustic imaging" Interface Focus. 2011 Aug 6; 1(4): 602–631.
5- Vasilis Ntziachristos "Going deeper than microscopy: the optical imaging frontier in biology", Nature Methods 7, 603–614 (2010)
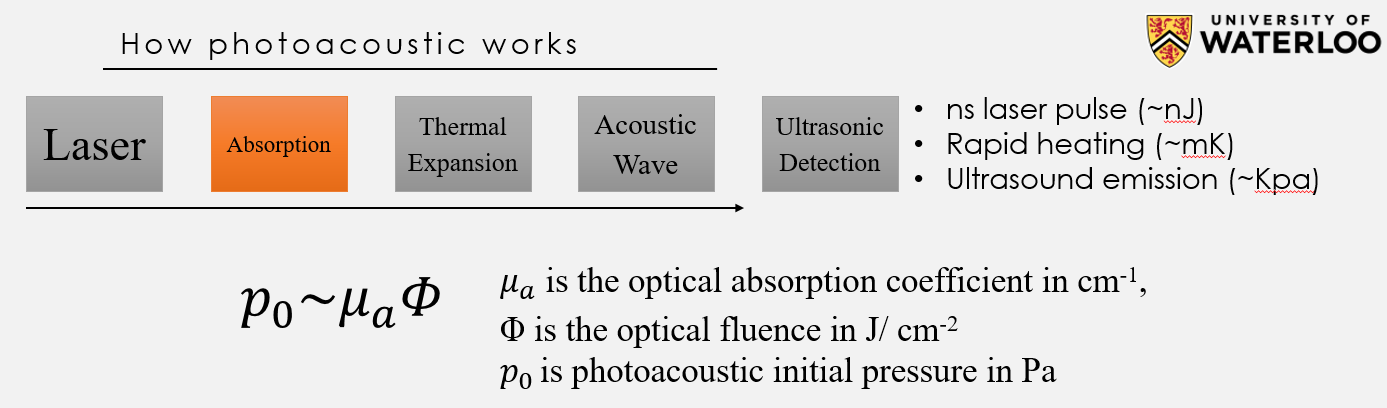
Photoacoustic Remote Sensing (PARS) microscopy
Similar to ultrasound imaging, traditional photoacoustic imaging systems require a coupling media, such as water or ultrasound gel, in between the ultrasound detector and the sample. This limits the use of these imaging systems in several clinical applications including wound healing, burn diagnostics, surgery, brain imaging, and many other endoscopic procedures where physical contact is impractical or undesirable as it may cause further damage or infection.There are also significant depth limitations to cellular-resolution, optically-focused, bio-microscopy which are hampering our ability to unravel cellular information at depths beyond ~1mm in living subjects. Development of a non-contact deep cellular-resolution PA is essential for many clinical and pre-clinical applications.
We recently pioneered a new technique (Photoacoustic Remote Sensing - PARS) that allows photoacoustic imaging without the need for any coupling media. This technique uses all-optical detection to remotely detect the photoacoustic signals through air and is capable of providing real-time imaging with cellular resolution. To achieve this, we use two laser beams at different wavelengths. One is visible light used to generate the signal, while the other is near-infrared invisible light used to detect the signal. The energy of these beams are very low and not harmful to tissue. PARS can provide breath-taking images of blood vessels down to the capillary level and can even visualize individual red blood cells in real-time. PARS is also capable of visualizing physiologically important parameters such as the oxygen saturation of blood vessels and can be used to distinguish arteries and veins. This is important for various disease including cancer, diabetes, and ischemia. We believe that this technology will have significant impact in visualizing and understanding cancer biology, as well, impacting dermatological application and even enabling functional brain imaging.
Similar to ultrasound imaging, traditional photoacoustic imaging systems require a coupling media, such as water or ultrasound gel, in between the ultrasound detector and the sample. This limits the use of these imaging systems in several clinical applications including wound healing, burn diagnostics, surgery, brain imaging, and many other endoscopic procedures where physical contact is impractical or undesirable as it may cause further damage or infection.There are also significant depth limitations to cellular-resolution, optically-focused, bio-microscopy which are hampering our ability to unravel cellular information at depths beyond ~1mm in living subjects. Development of a non-contact deep cellular-resolution PA is essential for many clinical and pre-clinical applications.
We recently pioneered a new technique (Photoacoustic Remote Sensing - PARS) that allows photoacoustic imaging without the need for any coupling media. This technique uses all-optical detection to remotely detect the photoacoustic signals through air and is capable of providing real-time imaging with cellular resolution. To achieve this, we use two laser beams at different wavelengths. One is visible light used to generate the signal, while the other is near-infrared invisible light used to detect the signal. The energy of these beams are very low and not harmful to tissue. PARS can provide breath-taking images of blood vessels down to the capillary level and can even visualize individual red blood cells in real-time. PARS is also capable of visualizing physiologically important parameters such as the oxygen saturation of blood vessels and can be used to distinguish arteries and veins. This is important for various disease including cancer, diabetes, and ischemia. We believe that this technology will have significant impact in visualizing and understanding cancer biology, as well, impacting dermatological application and even enabling functional brain imaging.
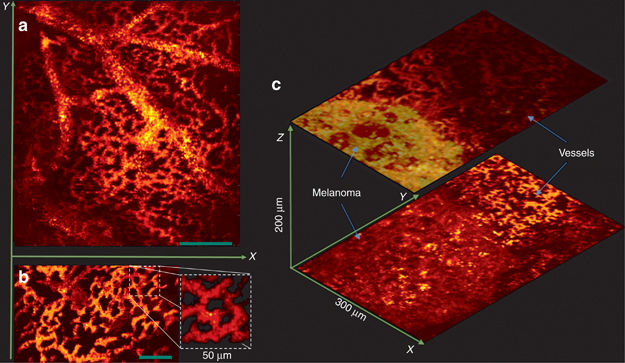
In vivo images of the CAM from a chicken embryo. (a) En-face C-scan PARS images (b) A snapshot of real-time imaging of capillaries at 30 FPS. (Media 1) (c) PARS images of a melanoma tumor and surrounding vasculature. Scale bar: 100 μm. Ref: Parsin Hajireza et. al, Non-interferometric photoacoustic remote sensing microscopy, Nature's Light: Science & Applications 6, e16278 (2017) https://www.nature.com/articles/lsa2016278
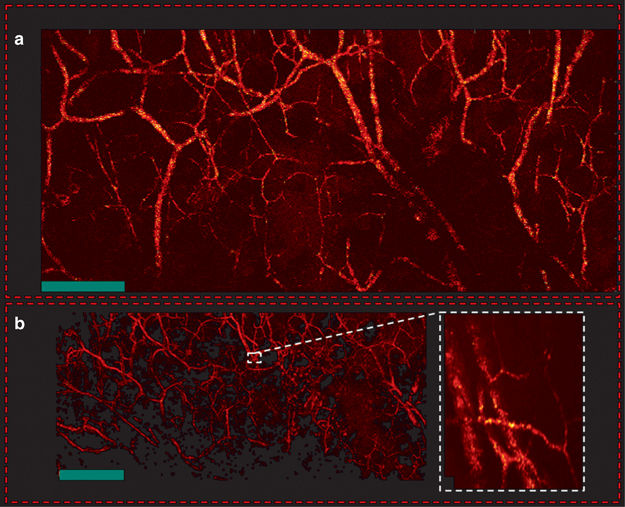
In vivo en-face mouse ear images. (a) PARS images using two-axis lateral mechanical scanning. (b) Larger field of view images using mechanical scanning as well as a zoomed in image of both capillary beds and larger blood vessels using fast scanning mirrors. Scale bar: 500 μm. Ref: Parsin Hajireza et. al, Non-interferometric photoacoustic remote sensing microscopy, Nature's Light: Science & Applications 6, e16278 (2017) https://www.nature.com/articles/lsa2016278
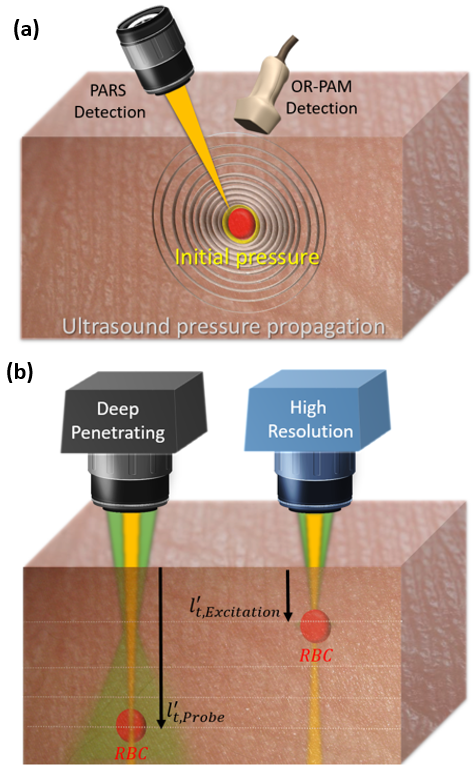
dPARS mechanism (a) PARS versus optical resolution photoacoustic microscopy (OR-PAM) detection mechanism. PARS technology, unlike any other photoacoustic imaging systems, does not measure propagated ultrasound pressures but rather the initial pressure generated at the origin. (b) A comparison between two different PARS modes, a deep penetrating mode and a high-resolution mode. RBC: red blood cell
Read more:
1- P. Hajireza, Wei Shi, Kevan Bell, Robert J. Paproski and Roger Zemp, “Non-Interferometric Photoacoustic Remote Sensing Microscopy” Nature’s Light: Science & Applications (2017) 6, e16278; doi: 10.1038/lsa.2016.278
2- P. Hajireza, Kevan Bell, Wei Shi, James Shapiro and Roger Zemp, Deep Non-Contact Photoacoustic Initial Pressure Imaging, Optica 2018.
3- Kevan L. Bell, Parsin Hajireza, Wei Shi, and Roger J. Zemp, "Temporal evolution of low-coherence reflectrometry signals in photoacoustic remote sensing microscopy," Appl. Opt. 56, 5172-5181 (2017).
4- Junjie Yao, “When pressure meets light: detecting the photoacoustic effect at the origin”, News and Views, Nature’s Light: Science & Applications (2017) 6, e17062; doi:10.1038/lsa.2017.62
5- Kevan Bell, Parsin Hajireza, and Roger Zemp, "Scattering cross-sectional modulation in photoacoustic remote sensing microscopy," Opt. Lett. 43, 146-149 (2018)
1- P. Hajireza, Wei Shi, Kevan Bell, Robert J. Paproski and Roger Zemp, “Non-Interferometric Photoacoustic Remote Sensing Microscopy” Nature’s Light: Science & Applications (2017) 6, e16278; doi: 10.1038/lsa.2016.278
2- P. Hajireza, Kevan Bell, Wei Shi, James Shapiro and Roger Zemp, Deep Non-Contact Photoacoustic Initial Pressure Imaging, Optica 2018.
3- Kevan L. Bell, Parsin Hajireza, Wei Shi, and Roger J. Zemp, "Temporal evolution of low-coherence reflectrometry signals in photoacoustic remote sensing microscopy," Appl. Opt. 56, 5172-5181 (2017).
4- Junjie Yao, “When pressure meets light: detecting the photoacoustic effect at the origin”, News and Views, Nature’s Light: Science & Applications (2017) 6, e17062; doi:10.1038/lsa.2017.62
5- Kevan Bell, Parsin Hajireza, and Roger Zemp, "Scattering cross-sectional modulation in photoacoustic remote sensing microscopy," Opt. Lett. 43, 146-149 (2018)

Copyright © 2018. All Rights Reserved.